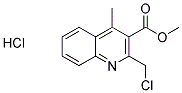
METHYL 2-(CHLOROMETHYL)-4-METHYLQUINOLINE-3-CARBOXYLATE HYDROCHLORIDE
methyl 2-(chloromethyl)-4-methylquinoline-3-carboxylate hydrochloride
CAS: 900641-03-4
Molecular Formula: C13H12ClNO2
METHYL 2-(CHLOROMETHYL)-4-METHYLQUINOLINE-3-CARBOXYLATE HYDROCHLORIDE - Names and Identifiers
METHYL 2-(CHLOROMETHYL)-4-METHYLQUINOLINE-3-CARBOXYLATE HYDROCHLORIDE - Physico-chemical Properties
| Molecular Formula | C13H12ClNO2 |
| Molar Mass | 249.69 |
| Storage Condition | Room Temprature |
METHYL 2-(CHLOROMETHYL)-4-METHYLQUINOLINE-3-CARBOXYLATE HYDROCHLORIDE - Risk and Safety
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| HS Code | 29334900 |
METHYL 2-(CHLOROMETHYL)-4-METHYLQUINOLINE-3-CARBOXYLATE HYDROCHLORIDE - Introduction
Methyl 2-(chloromethyl)-hydrochloride is an organic compound. The following is a brief description of its nature, use, preparation and safety information:
Nature:
-Appearance: Usually a white crystalline solid
-Molecular formula: C14H14ClNO2 · HCl
-Molecular weight: 321.19g/mol
-Melting point: about 198-202°C
-Solubility: Soluble in water, alcohol and ketone solvents
Use:
- methyl 2-(chloromethyl)-hydrochloride is often used as an intermediate in organic synthesis to prepare other organic compounds.
-It can also be used as a research reagent for drugs or drug precursors.
Method:
- methyl 2-(chloromethyl)-hydrochloride can be synthesized by the following steps:
1. First, 2-methyl quinoline -3-carboxylic acid methyl ester is reacted with anhydrous methylene chloride to generate 2-chloromethyl -4-methyl quinoline.
2. Next, 2-chloromethyl-4-methylquinoline is reacted with hydrochloric acid to obtain 4-methyl-2-chloromethylquinoline.
3. Finally, 4-methyl -2-chloromethylquinoline is reacted with methanol to generate 4-methyl -2-chloromethylquinoline -3-carboxylic acid methyl ester.
4. React 4-methyl -2-chloromethylquinoline -3-carboxylic acid methyl ester with hydrochloric acid to prepare methyl 2-(chloromethyl)-hydrochloride.
Safety Information:
- methyl 2-(chloromethyl)-hydrochloride specific risk factors may vary depending on the use and storage conditions, so the safety data sheet (SDS) provided by the supplier should be read carefully before use.
-During use or handling, appropriate laboratory safety procedures must be followed and appropriate personal protective measures must be taken, such as wearing chemical laboratory protective glasses, gloves and laboratory clothing.
-The compound is toxic and should be avoided by inhalation, ingestion or skin contact, and avoid contact with strong oxidants. After any contact, rinse immediately with plenty of water and seek medical help.
-When storing, it should be stored in a dry, low temperature, well-ventilated place, and away from fire and combustibles.
Nature:
-Appearance: Usually a white crystalline solid
-Molecular formula: C14H14ClNO2 · HCl
-Molecular weight: 321.19g/mol
-Melting point: about 198-202°C
-Solubility: Soluble in water, alcohol and ketone solvents
Use:
- methyl 2-(chloromethyl)-hydrochloride is often used as an intermediate in organic synthesis to prepare other organic compounds.
-It can also be used as a research reagent for drugs or drug precursors.
Method:
- methyl 2-(chloromethyl)-hydrochloride can be synthesized by the following steps:
1. First, 2-methyl quinoline -3-carboxylic acid methyl ester is reacted with anhydrous methylene chloride to generate 2-chloromethyl -4-methyl quinoline.
2. Next, 2-chloromethyl-4-methylquinoline is reacted with hydrochloric acid to obtain 4-methyl-2-chloromethylquinoline.
3. Finally, 4-methyl -2-chloromethylquinoline is reacted with methanol to generate 4-methyl -2-chloromethylquinoline -3-carboxylic acid methyl ester.
4. React 4-methyl -2-chloromethylquinoline -3-carboxylic acid methyl ester with hydrochloric acid to prepare methyl 2-(chloromethyl)-hydrochloride.
Safety Information:
- methyl 2-(chloromethyl)-hydrochloride specific risk factors may vary depending on the use and storage conditions, so the safety data sheet (SDS) provided by the supplier should be read carefully before use.
-During use or handling, appropriate laboratory safety procedures must be followed and appropriate personal protective measures must be taken, such as wearing chemical laboratory protective glasses, gloves and laboratory clothing.
-The compound is toxic and should be avoided by inhalation, ingestion or skin contact, and avoid contact with strong oxidants. After any contact, rinse immediately with plenty of water and seek medical help.
-When storing, it should be stored in a dry, low temperature, well-ventilated place, and away from fire and combustibles.
Last Update:2024-04-09 21:01:54
Supplier List
Product Name: METHYL 2-(CHLOROMETHYL)-4-METHYLQUINOLINE-3-CARBOXYLATE HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 900641-03-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 900641-03-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: METHYL 2-(CHLOROMETHYL)-4-METHYLQUINOLINE-3-CARBOXYLATE HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 900641-03-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 900641-03-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
